The rituals with hair are seen all over the world. In India, hair and its maintenance is probably still a time-consuming job for mothers with young girls and boys. I am still struck by little children going to school in India with shiny black hair neatly combed for boys and braided evenly for the girls as they trot to school. It does not matter how poor the family is, the children are very well-groomed.
My mother would spend everyday combing, oiling and braiding my long hair before we rushed off to school. Then, during the weekend, ‘oil bath’ and local powders called ‘Sheekapodi’ were used to clean the hair. The concept of shampoo was non-existent let alone entering hair salons. Today nail and hair salons proliferate not only in western countries, but in India as well.
We will look at the chemicals used in hair and nail salons and talk about the effects they have especially on the workers.

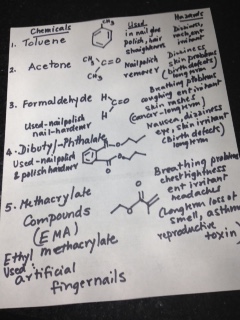

Toluene is used in nail gluing, nail polish and in hair straighteners. This organic compound is benzene with a methyl group attached. (Benzene is basically a cyclic compound with 6 carbons and 6 hydrogen atoms.) Short -term exposure may cause dizziness, rashes and ear-nose -throat (ENT) irritation. Acetone is a nail polish remover and is also available in any supermarket. This also causes dizziness, skin problems and long-term exposure leads to birth defects, if the workers are pregnant. Formaldehyde is used for nail polish as well as a nail-hardener: causes breathing problems, coughing , skin rashes and is an ENT irritant. Long-term exposure could lead to cancer. Another organic compound shown as #4 in the second figure, di-butyl phthalate, is used like formaldehyde and causes nausea, dizziness along with birth defects with long-term exposure. And then there is EMA, which is ethyl methacrylate (#5, Fig.2), used in artificial fingernails, that causes similar problems in workers. (Ref. 1,2)
Needless to say, I have looked at only a few chemicals in the nail salon industry. In addition, when it comes to hair salons, there are many products to worry about. A ‘perm’ or permanent was done at home by women who grew up in the ’50s. Women used keratin , a protein that actually weakens hair. (Ref. 3 and in Nuggets of Information) Weaving of hair is done not just in the African-American community, but now every celebrity/person with money who wants fuller, thicker hair can get it done. There are men , besides women, who are going in for hair extensions. The process involves stitching, gluing human hair to the scalp and can cause severe skin rashes, allergies and even loss of hair. These glues also affect the workers who inhale it all the time. (Refs.1,4,5) The glue contains styrene, an ethene attached to a benzene molecule. It causes dizziness, headaches and long- term exposure may lead to cancer. (Ref.1 )(Fig.3) Then there is the above-mentioned formaldehyde that is used in creating Brazilian Blowout, a hair treatment that will straighten curls and frizz or create it. We already know that it is toxic for the workers. Ref.6)
The list of toxic chemicals goes on and on. There is more information under Nuggets of Information. I have included some alternatives to the regular list of hair and nail salons. But we must look not just for our own health concerns but those of the workers who are constantly exposed to this barrage of toxicity. The good news is that awareness is growing, there is more advocacy for the workers, but all that is being done is just a drop in the bucket. More needs to be done to change this love affair we have with the dead cells in our bodies.
Activities for Middle School Teachers:
Talk about hair and nail grooming practices in different parts of the world. Students will learn geography and culture all over the world.
Study the history of African-American hair and special hair salons for them.
Go to the supermarket and check out the hair and nail products division and study the chemical ingredients in all of them. Let students do a presentation on the chemical structure, usage of product and its toxic side-effects.
Study hair styles through the ages especially in the United States focusing on Caucasian and African American groups.
Nuggets of Information:
Human hair export, from India, is a multi-million dollar industry. Indians, especially Hindus, believe in shaving off all their hair as a gift to the gods. Men and women with long black hair, frequently go to temples to be shorn of their tresses. The demand for wigs and hair extensions is high in the US, UK and parts of Europe. (Ref. 7)
Nails and hair are considered extensions of the skin. Hair and nails cells grow at the base of the root, and when they move upward, they are cut off from nourishment to form a hard protein called keratin. This keratin and the dead cells form the shaft of hair or nail. (Ref.8)
Four out of ten workers in the nail and salon industry are poor Asians with limited English-speaking skills, uninsured and working in badly-ventilated spaces. Fortunately, ACLU and other labor organizations are advocating for their awareness of a safe environment to work in. The California Health Nail Salon Collaborative, along with environmental and community organizations are trying to ban the ‘toxic trio’ of formaldehyde, dibutyl-phthalate and toluene in hair/nail salons. (Ref.7)
Celebrities like Jennifer Anniston have come out openly to talk about the hazards and adverse effects of hair extensions. Some famous singers have had bald patches in their hair because of these extensions. Perhaps the public will not be swayed and charmed by the commercials that laud their beauty. (Ref.9)
Recently, there is an increase in American NFL football players with long dreadlocks. Some of them could be wearing hair extensions, since they are getting popular among men now. (Ref. 10)
How does a ‘perm’ work? The protein molecules in the hair called keratin are arranged in straight molecules joined by disulfide bonds (-S-S-) which gives strength to the hair. (Actually the amino acid cystein in the keratin molecule forms the -S-S- bond with another cystein in a second keratin molecule.) Ammonium thioglycolate, ( HSCH2CO2NH4) or ‘thiol’ can break the bonds and allow manipulation of hair: either put in curlers to curl straight hair or use a flat iron to straighten curly hair. This is a reduction process (which is opposite of oxidation) and oxygen is lost or hydrogen is added. Following this Hydrogen peroxide, H2O2 , is added to strengthen the hair, an oxidation process.
Sometimes, lye soap is used instead of ‘thiol’. Lye is a 10% solution of NaOH or sodium hydroxide which does the same reduction; then it is thoroughly washed out with conditioner and water to prevent corrosion of the scalp. (Ref.11) As you can see, continuous perming of hair can lead to seriously damaged hair.
Some encouraging news to help us modify our nail and hair salon addictions:
- Jenna Hipp has come up with nail polishes that avoid the toxic- trio chemicals, check out her other products. (Ref.12)
- Campaign for Safe Cosmetics is an advocacy group that helps workers and consumers make smart choices and are aware of the hazards and side effects of products that keep coming out in the cosmetics industry. (Ref.13)
- No more dirty looks is a blog site with good alternatives for hair products. Check out their book as well. (Ref. 14)
References:
1.http://www.womensvoices.org/avoid-toxic-chemicals/salon-products/toxic-chemicals-in-salon-products-workers/
2.http://www.scientificamerican.com/article/these-4-chemicals-may-pose-the-most-risk-for-nail-salon-workers/
3.http://www.ask.com/beauty-fashion/chemicals-used-perming-hair-79b66ed09938762a
4.http://www.consumerreports.org/cro/2013/02/the-dangers-of-hair-extensions/index.htm
5.http://www.nydailynews.com/life-style/hair-extensions-latest-well-tressed-man-article-1.1497062
6.http://www.alternet.org/story/155982/toxic_chemicals_in_hair_and_na il_salons_create_serious_suffering_in_the_name_of_beauty/
7.http://www.scienceandnature.org/IJEMS-Vol2(4)-Oct2011/IJEMS_V2(4)4.pdf
8.http://kidshealth.org/teen/your_body/body_basics/skin_hair_nails.ht ml
9.http://www.dailymail.co.uk/femail/article-2274414/Dangers-hair-extensions-Blinding-headaches-bleeding-scalps-permanent-baldness.html
10.http://www.nydailynews.com/life-style/hair-extensions-latest-well-tressed-man-article-1.1497062
11.http://www.humantouchofchemistry.com/the-chemistry-of-perming-rebonding.htm
12.http://www.therecessionista.com/2014/07/jenna-hipp-introduces-eco-friendly-nail-polish.html
13.http://www.safecosmetics.org/
14.O’Conner, Siobhan & Spunt, Alexandra, No More Dirty Looks (De Capo Lifelong Books, 2010)