When my mother was young, the perfume that young women of her time would crave for was Chanel No. 5! I remember my sister getting her first whiff of this exquisite perfume at a wealthy girlfriend’s house and raving about it.
Aldehydes played a quiet role in creating that popular perfume. (Ref. 1). Today we shall study aldehydes and ketones and note their structure and everyday uses.
Let us start with the simplest aldehyde: commonly known as formaldehyde,but the IUPAC name is methanal. Note the ‘al’ ending, whereas for alcohols, the ending is ‘ol’. All aldehydes have H-C=O structure, so the first aldehyde is shown with an additional H or hydrogen atom. The next aldehyde, ethanal or acetaldehyde has a methyl group instead of the H, or hydrogen atom. Fig. 1 has the first four in the series. (Ref. 2)
Ketones are very similar to aldehydes, but they do not have the H-C=O structure; only the C=O structure, or the carbonyl structure. The carbon atom could have two methyl groups and then could be more carbon atoms. Again, Fig.1 has the first four in the ketone series. (Ref. 2)
Since aldehydes have the hydrogen present, they are not as stable as ketones and can be oxidized. Since ketones do not have that hydrogen atom, they are resistant to oxidation. (Ref. 3)
The carbonyl group,C=O,is characteristic of both aldehydes and ketones. The oxygen atom is far more electronegative than the carbon atom and the presence of the extra bond (double bond) increases the pull of the electrons towards the O. The high polarity of this bond in aldehydes and ketones results in higher boiling points relative to comparative hydrocarbons. (Ref. 3)
Smaller molecules of aldehydes and ketones are soluble in water; they hydrogen bond with water molecules. As chain length increases, the hydrocarbon ‘tails'(all hydrocarbon bits apart from the carbonyl group) of the molecule come in the way of hydrogen bonding. (Ref. 3)
Uses of aldehydes and ketones: Formaldehyde and acetaldehyde (methanal and ethanal) are used extensively in industry,resins. Butyraldehyde or butanal,is used as a plasticizer. Higher aldehydes are used as deodorants, favors and detergents. (Ref. 3)
Ketones are used mainly in pharmaceuticals,polymers, solvents. Ketones play a significant role in biochemistry. (Ref. 3)
So once again, organic compounds play a vital role in our daily lives.
Activities for Middle School Teachers:
Let students write straight and branched carbon groups for aldehydes and ketones for the first ten in the series. How many isomers can you get? Are there any enantiomers, stereo- isomers? Name them all using the IUPAC nomenclature.
Nuggets of Information:
Many aldehydes are derived from the de- hydrogenation of corresponding alcohols, hence the name ‘ al-dehyde’. (Ref. 4).
Certain foods and flavors in our diet contain aldehydes. Cinnamon, almonds and vanilla are examples of aldehydes cinnamaldehyde, benzaldehyde and vanillin respectively. (Ref. 5). All these compounds have a benzene ring.
Aldehydes and ketones play an important role in the chemistry of carbohydrates. A carbohydrate is literally a hydrate of carbon. The sugars, glucose and fructose are carbohydrates. Glucose is an aldehyde and fructose is a ketone. (Ref. 5)
When the body uses fat for energy,ketones are generated during fat metabolism. The fat gets converted to A T P. Ketones are produced as part of the process. When people eat less carbohydrate,the bodies turn to fat for energy, naturally more ketones are generated then. This is called ketosis. ( Ref.6)
Most cells use ketones for at least part of their energy. (Ref.6)
Ketones and aldehydes exhibit tautomerism: this is a special kind of isomerism, where the carbonyl group, C=O, converts to the hydroxyl group,OH group and a double bond. This is called keto-enol tautomerism. The u-tube illustrates it. (Ref. 7)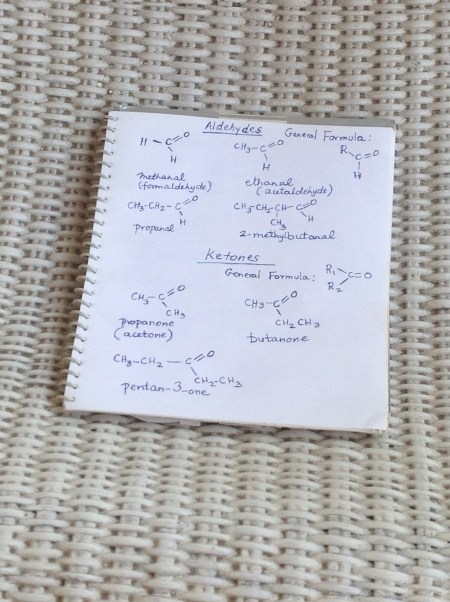
References
1. perfume shrine.blogspit.in
2. chemguide.co.uk
3. livewebtutors.com
4. brittanica.com
5. chemed.chem.perdue.edu
6. lowcarbdiets.about.com
7.u-tube, keto-enol tautomerism, Khan Academy
Tags: Aldehydes, flavorings, ketones, perfumes
Leave a comment